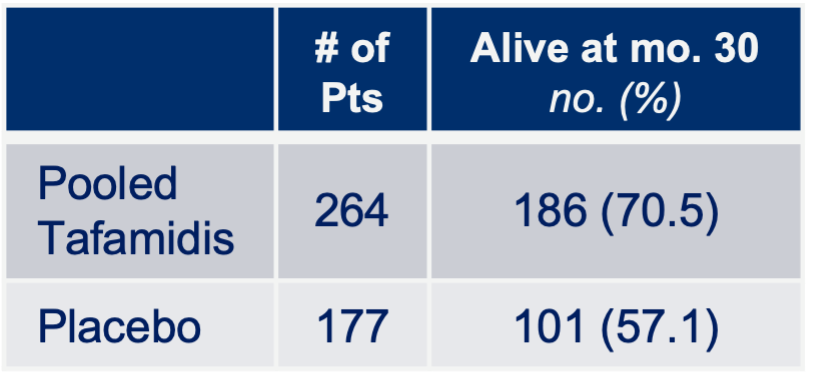
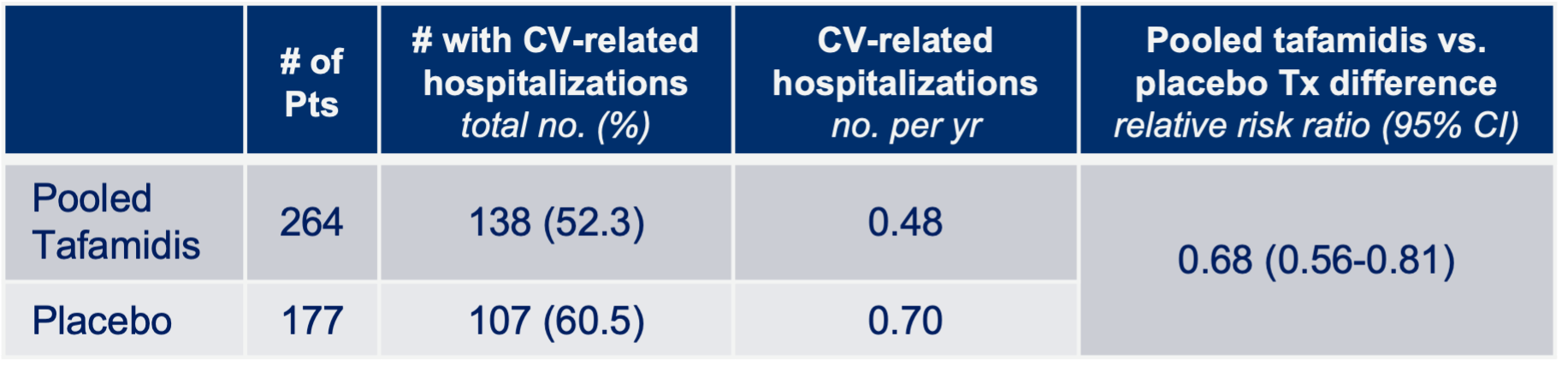
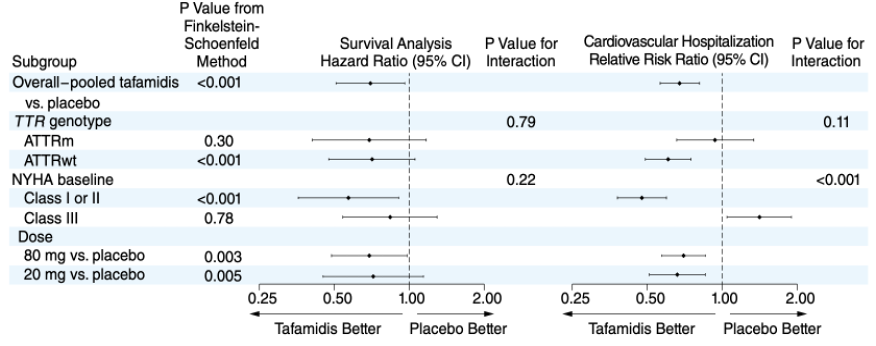
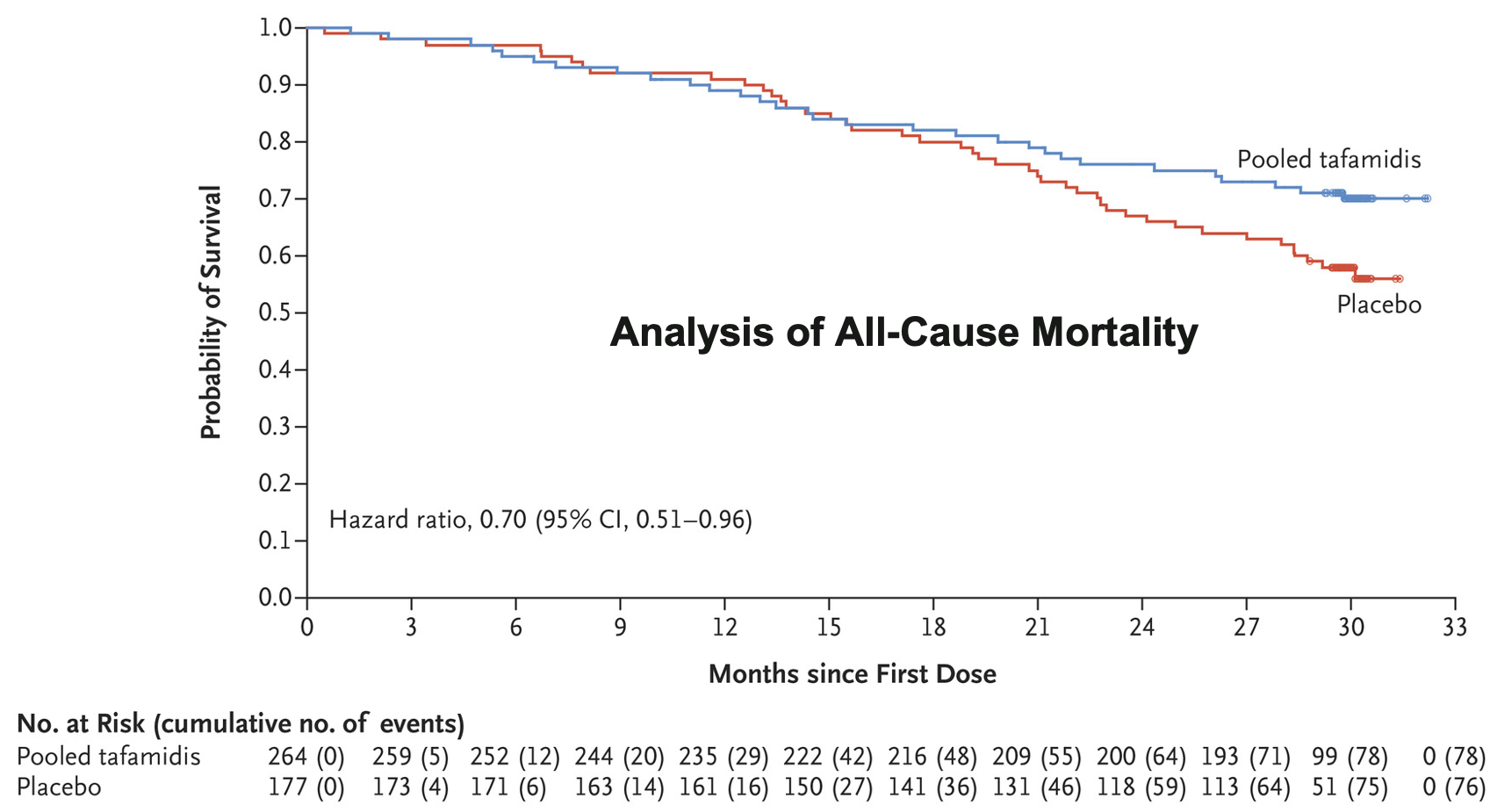
Notes:
* Mathew Maurer, MD (Columbia University Irving Medical Center, New York, NY), who published his views along with Jerry Gurwitz, MD (University of Massachusetts Medical School, Worcester), January 8, 2020, in JAMA Cardiology; Rodney Falk, MD (Brigham and Women’s Hospital, Boston, MA), who runs a specialized cardiac amyloidosis program; Ronald Witteles, MD (Stanford University Medical Center, CA), who is co-director of the amyloid program at his institution
Notes: ** Pfizer estimates the prevalence of ATTR-CM to be approximately 100,000 people in the United States and that approximately 4 to 5% of people are diagnosed, but Maurer and Gurwitz highlight one study suggesting a prevalence of 13% among patients with heart failure with preserved ejection fraction (HFpEF) and increased wall thickness.
Notes: *** OOP costs for most patients are difficult to pin down given rebates and subsidies, but researchers estimate patients pay roughly $1,000 to $2,000 per month if they don’t receive additional financial support.” – Maurer, MD
Notes: In cardiology, the makers of the PCSK9 inhibitors faced significant backlash when they initially priced alirocumab (Praluent; Sanofi/Regeneron) and evolocumab (Repatha; Amgen) at nearly $15,000 per year, although that drug class is for a much more common disease affecting a significantly larger number of patients. The companies eventually cut their prices—slashing the costs by roughly 60%—when uptake stalled.
Notes: ^ Two agents, inotersen (Tegsedi; Akcea) and patisiran (Onpattro; Alnylam), both of which are approved as orphan agents for the treatment of the much-rarer hereditary amyloidosis, a disease the primarily affects the nervous system, will be tested in patients with ATTR-CM. Patisiran currently has a list price of $350,000 per year, while inotersen costs $450,000. The author notes AG10 is an investigational small molecule TTR therapy being developed for and tested in cardiomyopathy and polyneuropathy, and it has already been designated an orphan drug by the FDA